Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Okazaki, Hiroyuki*; Idesaki, Akira*; Koshikawa, Hiroshi*; Matsumura, Daiju; Ikeda, Takashi*; Yamamoto, Shunya*; Yamaki, Tetsuya*
Journal of Physical Chemistry C, 127(49), p.23628 - 23633, 2023/12
Times Cited Count:0 Percentile:0(Chemistry, Physical)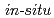 high energy resolution XAFS
high energy resolution XAFSYamamoto, Naoki*; Matsumura, Daiju; Hagihara, Yuto*; Tanaka, Kei*; Hasegawa, Yuta*; Ishii, Kenji*; Tanaka, Hirohisa*
Journal of Power Sources, 557, p.232508_1 - 232508_10, 2023/02
Times Cited Count:2 Percentile:26.88(Chemistry, Physical)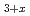 (M = Gd, Np) by the reaction of MN with Pd and chlorination of MPd
(M = Gd, Np) by the reaction of MN with Pd and chlorination of MPd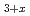 using cadmium chloride
using cadmium chlorideHayashi, Hirokazu; Shibata, Hiroki; Sato, Takumi; Otobe, Haruyoshi
Journal of Radioanalytical and Nuclear Chemistry, 332(2), p.503 - 510, 2023/02
Times Cited Count:0 Percentile:0.01(Chemistry, Analytical)The formation of MPd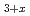 (M = Gd, Np) by the reaction of MN with Pd at 1323 K in Ar gas flow was observed. Cubic AuCu
(M = Gd, Np) by the reaction of MN with Pd at 1323 K in Ar gas flow was observed. Cubic AuCu -type GdPd
-type GdPd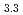 (
(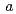 = 0.4081
= 0.4081  0.0001 nm) and NpPd
0.0001 nm) and NpPd (
(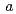 = 0.4081
= 0.4081  0.0001 nm) were identified, respectively. The product obtained from the reaction of NpN with Pd contained additional phases including the hexagonal TiNi
0.0001 nm) were identified, respectively. The product obtained from the reaction of NpN with Pd contained additional phases including the hexagonal TiNi -type NpPd
-type NpPd . Chlorination of the MPd
. Chlorination of the MPd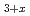 (M = Gd, Np) samples was accomplished by the solid-state reaction using cadmium chloride at 673 K in a dynamic vacuum. Pd-rich solid solution phase saturated with Cd and an intermetallic compound PdCd were obtained as by-products of MCl
(M = Gd, Np) samples was accomplished by the solid-state reaction using cadmium chloride at 673 K in a dynamic vacuum. Pd-rich solid solution phase saturated with Cd and an intermetallic compound PdCd were obtained as by-products of MCl formation.
formation.
Liu, B.*; Feng, R.*; Busch, M.*; Wang, S.*; Wu, H.*; Liu, P.*; Gu, J.*; Bahadoran, A.*; Matsumura, Daiju; Tsuji, Takuya; et al.
ACS Nano, 16(9), p.14121 - 14133, 2022/09
Times Cited Count:51 Percentile:98.66(Chemistry, Multidisciplinary)Ueda, Yuki; Eguchi, Ayano; Tokunaga, Kohei; Kikuchi, Kei*; Sugita, Tsuyoshi; Okamura, Hiroyuki; Naganawa, Hirochika
Industrial & Engineering Chemistry Research, 61(19), p.6640 - 6649, 2022/05
Times Cited Count:1 Percentile:11.64(Engineering, Chemical)no abstracts in English
Kimata, Tetsuya*; Kakitani, Kenta*; Yamamoto, Shunya*; Shimoyama, Iwao; Matsumura, Daiju; Iwase, Akihiro*; Mao, W.*; Kobayashi, Tomohiro*; Yamaki, Tetsuya*; Terai, Takayuki*
Physical Review Materials (Internet), 6(3), p.035801_1 - 035801_7, 2022/03
Times Cited Count:7 Percentile:68.86(Materials Science, Multidisciplinary)Ito, Tatsuya; Osugi, Haruka*; Osawa, Naoki*; Takahashi, Tadayuki*; Kim, S.-Y.*; Nagaishi, Ryuji
Analytical Sciences, 38(1), p.91 - 97, 2022/01
Times Cited Count:2 Percentile:29.37(Chemistry, Analytical)A novel ionic liquid (IL) functionalized with thiodiglycol amic acid containing a soft S donor was synthesized for the effective and efficient extraction of platinum group metals (Ru, Rh, and Pd) from aqueous nitric acid solutions, such as high-level radioactive liquid waste (HLLW). The IL allowed a rapid extraction of Pd(II) with an extraction ratio of approximately 100%. The extractions of Ru(III) and Rh(III) by the IL were slower than that of Pd(II), but the rates were accelerated by temperature elevation. The extractions of Ru(III) and Rh(III) at 50 C reached equilibrium within 4 and 8 h, respectively, with the extraction ratios of over 90% without assisting agents or other methods for the extraction system. Furthermore, the IL could extract more than 90% of Ru(III), Rh(III), and Pd(II) from the simulated HLLW within 2 h at 50
C reached equilibrium within 4 and 8 h, respectively, with the extraction ratios of over 90% without assisting agents or other methods for the extraction system. Furthermore, the IL could extract more than 90% of Ru(III), Rh(III), and Pd(II) from the simulated HLLW within 2 h at 50 C.
C.
 ]
] from hydrochloric acid using a simple urea extractant
from hydrochloric acid using a simple urea extractantUeda, Yuki; Morisada, Shintaro*; Kawakita, Hidetaka*; Wenzel, M.*; Weigand, J. J.*; Oto, Keisuke*
Separation and Purification Technology, 277, p.119456_1 - 119456_8, 2021/12
Times Cited Count:5 Percentile:29.15(Engineering, Chemical)no abstracts in English
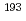 Ir M
Ir M ssbauer spectroscopic parameters of Vaska's complexes and their oxidative adducts
ssbauer spectroscopic parameters of Vaska's complexes and their oxidative adductsKaneko, Masashi; Nakashima, Satoru*
Inorganic Chemistry, 60(17), p.12740 - 12752, 2021/09
Times Cited Count:3 Percentile:31.46(Chemistry, Inorganic & Nuclear)In the present study, density functional theory (DFT) calculation was applied to Vaska's complexes of formula 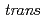 -[IrCl(CO)(PPh
-[IrCl(CO)(PPh )
) ], and their oxidative adducts with small molecules (YZ) including H
], and their oxidative adducts with small molecules (YZ) including H , i.e.,
, i.e., 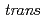 -[IrCl(YZ)(CO))(PPh
-[IrCl(YZ)(CO))(PPh )
) ], to successfully correlate the electronic states of the complexes with the corresponding
], to successfully correlate the electronic states of the complexes with the corresponding 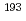 Ir M
Ir M ssbauer spectroscopic parameters. After confirming the reproducibility of the DFT methods for elucidating the equilibrium structures and
ssbauer spectroscopic parameters. After confirming the reproducibility of the DFT methods for elucidating the equilibrium structures and 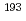 Ir M
Ir M ssbauer isomer shifts of the octahedral Ir complexes, the isomer shifts and quadrupole splitting values of Vaska's complexes and their oxidative adducts were calculated. A bond critical point analysis revealed that the tendency in the isomer shifts was correlated with the strength of the covalent interaction in the coordination bonds. In an electric field gradient (EFG) analysis of the oxidative adducts, the sign of the principal axis was found to be positive for the complex with YZ = Cl
ssbauer isomer shifts of the octahedral Ir complexes, the isomer shifts and quadrupole splitting values of Vaska's complexes and their oxidative adducts were calculated. A bond critical point analysis revealed that the tendency in the isomer shifts was correlated with the strength of the covalent interaction in the coordination bonds. In an electric field gradient (EFG) analysis of the oxidative adducts, the sign of the principal axis was found to be positive for the complex with YZ = Cl and negative for the complex with YZ = H
and negative for the complex with YZ = H . This reversal of the sign of the EFG principal axis was caused by the difference in the electron density distribution for the coordination bonds between Ir and YZ, according to a density of states analysis.
. This reversal of the sign of the EFG principal axis was caused by the difference in the electron density distribution for the coordination bonds between Ir and YZ, according to a density of states analysis.
Ueda, Yuki; Morisada, Shintaro*; Kawakita, Hidetaka*; Oto, Keisuke*
Separations (Internet), 8(9), p.139_1 - 139_15, 2021/09
Times Cited Count:9 Percentile:62.42(Chemistry, Analytical)no abstracts in English
Mishima, Ria; Inaba, Yusuke*; Tachioka, Sotaro*; Harigai, Miki*; Watanabe, Shinta*; Onoe, Jun*; Nakase, Masahiko*; Matsumura, Tatsuro; Takeshita, Kenji*
Chemistry Letters, 49(1), p.83 - 86, 2020/01
Times Cited Count:4 Percentile:20.61(Chemistry, Multidisciplinary)Separation of platinum group metals (PGMs) from high-level liquid waste generated from the reprocessing of spent nuclear fuels is important to produce good quality vitrified glass for final disposal. A new sorbent, Aluminum hexacyanoferrate (AlHCF), was synthesized and the general sorption behavior of PGMs from concentrated nitric acid was examined. Nitric acid caused substantial elution of AlHCF but the sorption of Pd stabilized the structure. Consequently, Rh was sorbed in the presence of Pd, whereas single Rh sorption caused complete dissolution of AlHCF. Relation between sorbed mount of Pd vs eluted Al and Fe revealed that the elution ratio of Al and Fe was not the same as molar ratio of synthesized AlHCF, indicating the re-sorption of Fe resulted in formation of new structure. The sorption mechanism of PGMs by this new sorbent, AlHCF, not only the simple ion exchange, but also oxidation reduction reaction as well as kinetics play important rule. Understanding the general sorption and dissolution behavior will help improve the sorption performance of PGMs by AlHCF.
Yasuda, Satoshi; Uchibori, Yosuke*; Wakeshima, Makoto*; Hinatsu, Yukio*; Ogawa, Hiroaki; Yano, Masahiro; Asaoka, Hidehito
RSC Advances (Internet), 8(66), p.37600 - 37605, 2018/11
Times Cited Count:12 Percentile:42.2(Chemistry, Multidisciplinary)We present a quantitative study on the effect of a newly obtained thermal history on the formation of Fe-N-C catalytic sites. A short and repeated heating process is employed as the new thermal history, where short heating (1 min) followed by quenching is applied to a sample with arbitrary repetition. Through electrochemical quantitative analysis, it is found that the new process effectively increases the Fe-N-C mass-based site density (MSD) to almost twice that achieved using a conventional continuous heating process, while the turn-over frequency (TOF) is independent of the process. Elemental analysis shows that the new process effectively suppresses the thermal desorption of Fe and N atoms during the initial formation stage and consequently contributes to an increase in the Fe-N-C site density. The resultant catalytic activity (gravimetric kinetic current density (0.8 V vs. RHE)) is 1.8 times higher than that achieved with the continuous heating process.
Cui, Y.-T.*; Harada, Yoshihisa*; Niwa, Hideharu*; Oshima, Masaharu*; Hatanaka, Tatsuya*; Nakamura, Naoki*; Ando, Masaki*; Yoshida, Toshihiko*; Ishii, Kenji*; Matsumura, Daiju
NanotechJapan Bulletin (Internet), 11(4), 6 Pages, 2018/08
no abstracts in English
Saeki, Morihisa*; Asai, Shiho; Oba, Hironori*
Bunseki, 2018(4), p.138 - 143, 2018/04
Platinum group metal (PGM) has attracted much attention in light of increasing demands in the industrial sector. A wide variety of techniques specialized for PGM separation, such as, solvent extraction, solid phase extraction, and molten salt electrolysis have been developed so far. Among such techniques, a newly developed separation technique based on laser-induced particulate formation can be a promising alternative to conventional ones. It enables non-contact and highly-selective separation with a simple operation. In this review, the research history and the basic mechanism of laser-induced particulate formation were outlined. Several applications were also mentioned, focusing on our latest research progress which achieved a world first quantitation of radioactive palladium in a spent nuclear fuel sample.
Onishi, Takashi; Sekioka, Ken*; Suto, Mitsuo*; Tanaka, Kosuke; Koyama, Shinichi; Inaba, Yusuke*; Takahashi, Hideharu*; Harigai, Miki*; Takeshita, Kenji*
Energy Procedia, 131, p.151 - 156, 2017/12
Times Cited Count:11 Percentile:98.82(Energy & Fuels)no abstracts in English
Okamoto, Yoshihiro; Nagai, Takayuki; Shiwaku, Hideaki; Inose, Takehiko*; Sato, Seiichi*
Nihon Genshiryoku Gakkai Wabun Rombunshi, 16(4), p.180 - 190, 2017/12
X-ray absorption fine structure (XAFS) and imaging XAFS analyses were performed to elucidate chemical state of rhodium in the simulated waste glass. The chemical forms of Rh in the glass were evaluated to be 84% RhO and 16% metal/alloy as the result of linear combination analysis of EXAFS data. According to the imaging XAFS analysis, the chemical form of Rh which was located together with Ru was mainly oxide (RhO
and 16% metal/alloy as the result of linear combination analysis of EXAFS data. According to the imaging XAFS analysis, the chemical form of Rh which was located together with Ru was mainly oxide (RhO ). It suggests that stable (Ru,Rh)O
). It suggests that stable (Ru,Rh)O solid solution exists in the simulated glass. On the other hand, that of Rh of which distribution did not accord with Ru in the glass was mainly metallic. In the case of metallic Rh in the glass, it tended to become an aggregation form. It can be concluded that the chemical state of Rh was much affected by the existence and distribution of Ru element.
solid solution exists in the simulated glass. On the other hand, that of Rh of which distribution did not accord with Ru in the glass was mainly metallic. In the case of metallic Rh in the glass, it tended to become an aggregation form. It can be concluded that the chemical state of Rh was much affected by the existence and distribution of Ru element.
Narita, Hirokazu*; Suzuki, Tomoya*; Motokawa, Ryuhei
Nihon Kinzoku Gakkai-Shi, 81(4), p.157 - 167, 2017/04
Times Cited Count:17 Percentile:62.47(Metallurgy & Metallurgical Engineering)Maeda, Motoki*; Narita, Hirokazu*; Tokoro, Chiharu*; Tanaka, Mikiya*; Motokawa, Ryuhei; Shiwaku, Hideaki; Yaita, Tsuyoshi
Separation and Purification Technology, 177, p.176 - 181, 2017/04
Times Cited Count:22 Percentile:58.37(Engineering, Chemical)Suzuki, Tomoya; Morita, Keisuke; Sasaki, Yuji; Matsumura, Tatsuro
Separation Science and Technology, 51(17), p.2815 - 2822, 2016/09
Times Cited Count:8 Percentile:26.31(Chemistry, Multidisciplinary)To understand the adsorption properties of styrene-divinylbenzene copolymer functionalized with 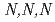 -trimethylglycine, AMP03, the adsorption behaviors for platinoid ions (Ru(III), Rh(III), and Pd(II)) were examined. Furthermore, we performed adsorption experiments using sample solutions with adding triethylamine, thiourea, and
-trimethylglycine, AMP03, the adsorption behaviors for platinoid ions (Ru(III), Rh(III), and Pd(II)) were examined. Furthermore, we performed adsorption experiments using sample solutions with adding triethylamine, thiourea, and 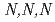 -trimethylglycine. Based on the adsorption data obtained in this study, we performed chromatographic experiments. The results indicated that all platinoid ions in the feed solution completely adsorbed on AMP03, and almost 80% of the adsorbed platinoid ions were recovered. These results show that AMP03 has the potential to recover Ru(III), Rh(III), and Pd(II) from high-level liquid waste.
-trimethylglycine. Based on the adsorption data obtained in this study, we performed chromatographic experiments. The results indicated that all platinoid ions in the feed solution completely adsorbed on AMP03, and almost 80% of the adsorbed platinoid ions were recovered. These results show that AMP03 has the potential to recover Ru(III), Rh(III), and Pd(II) from high-level liquid waste.
Yamagishi, Isao; Odakura, Makoto; Ichige, Yoshiaki; Kuroha, Mitsuhiko; Takano, Masahide; Akabori, Mitsuo; Yoshioka, Masahiro*
Proceedings of 21st International Conference & Exhibition; Nuclear Fuel Cycle for a Low-Carbon Future (GLOBAL 2015) (USB Flash Drive), p.1113 - 1119, 2015/09
The characteristics of insoluble residues in fine suspension at the Rokkasho Reprocessing Plant were analyzed. The insoluble residues were washed with oxalic acid solution to dissolve zirconium molybdate residues. XRD profiles of unwashed residues showed the presence of a noble metal alloy, zirconium molybdate, and zirconia, but zirconium molybdate was not found after washing. More than 50% of the Sb-125 and Pu in thee residues was washed out as well. The noble metal alloy composed of Mo, Tc, Ru, Rh, and Pd occupied more than 90% of the total weight of 12 elements (Ca, Cr, Fe, Ni, Zr, Mo, Tc, Ru, Rh, Pd, Te, and U) found in the residues. In consideration of the chemical forms of 12 elements, the alloy-to-residue weight ratio was evaluated to be 64% and 78% with and without 18% of an unknown component, respectively.